The hydrogen in a particular hydrogen gas thermometer has a volume of 150.0 cm 3 when immersed in a mixture of ice and water (0.00 C). Note: For these types of question we should have a clear understanding of different gas laws that is Boyle’s law, Charles’s law, Gay-Lussac’s law, combined gas law, Avogadro’s law, and Ideal gas law. Temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as the temperature changes at constant pressure.
Using this concept we can easily estimate the temperature by calculating the volume of gas at a specified temperature by using the formula

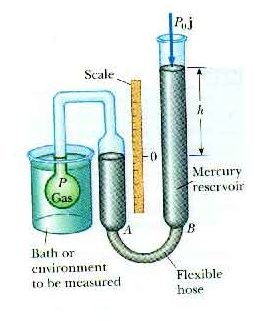
Charles's Law states that if the temperature of a gas increases, its volume also increases. We all know that a gas thermometer works on the principle given by Charles's Law. As we have seen that a constant-volume gas thermometer is filled with ideal gas which can be achieved in high temperature and low pressure because in these conditions there is almost zero interaction between the gas molecules. A Constant -volume gas thermometer provides accurate measurement over a wide range of values. Liquid Pressure Thermometer: Usually Mercury is used as liquid Principle of working: expansion of liquid due to an increase in the pressure in a given volume Range :-30C to 550C 10. This is because when the temperature is decreased the pressure is decreased and similarly as the temperature is increased the pressure is increased. Advantages of constant volume Gas thermometer:-) As the expansion of gas is large, so the Gas thermometer are very sensitive. Therefore, the ideal gas temperature scale is identical to the Kelvin scale as long as the gas in the bulb does not condense or dissociate. Ingress protection (IP Code): IP55 or IP65.Now we measure the pressure of the gas which can be used as a measure of temperature. Immersion stem diameters: Ø6, Ø8, Ø9, Ø12 mm A constant-volume gas thermometer is used to determine the value of absolute zero in degrees Celsius by plotting Temperature vs. Dial case diameters: Ø63, Ø80, Ø100, Ø160, Ø200, Ø250 mm The meaning of gas thermometer is a thermometer containing gas (as hydrogen) as the enclosed thermometric substance, variations in temperature being indicated by the change in pressure of a fixed quantity of gas required to maintain the gas at a constant volume or the change in volume of a fixed quantity of gas maintained at a constant pressure. When the thermometer is in contact with water at its triple point (273.16 K) the pressure in the thermometer is 8.500 × 104 Pa. Specially customised versions, on demand.
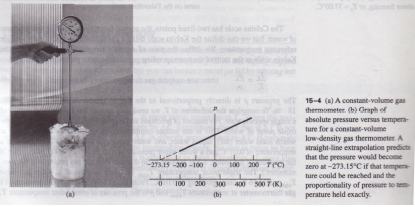
Mechanical engineering and apparatus manufacture.Energy saving and preservation, process engineering.compliance with the highest demands of measuring technique Showing that at constant pressure thermometer is used to determine the temperature of pressure versus temperature for a gas.

